|
Although tabulated values are usually corrected to 298 K, that correction is often smaller than the uncertainty in the measured value. The enthalpy of vaporization is often quoted for the normal boiling temperature of the substance. The enthalpy of vaporization is a function of the pressure at which the transformation ( vaporization or evaporation) takes place. 
In thermodynamics, the enthalpy of vaporization (symbol ∆ H vap), also known as the ( latent) heat of vaporization or heat of evaporation, is the amount of energy ( enthalpy) that must be added to a liquid substance to transform a quantity of that substance into a gas. Temperature-dependency of the heats of vaporization for water, methanol, benzene, and acetone ( March 2016) ( Learn how and when to remove this template message) Please help to improve this article by introducing more precise citations. 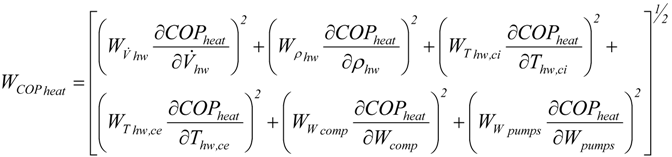 
This article includes a list of general references, but it lacks sufficient corresponding inline citations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
